How Long Did the Pfizer COVID Vaccine Trial Last?
Let's just say, they should have used Pfizer's little blue pill.
How long did the Pfizer/BioNTech COVID vaccine clinical trial last?
The short answer is: 97 days.
Here’s the long answer:
When unblinding began in earnest on December 14, 2020 — with the FDA’s blessing 3 days after they granted an EUA — the trial subjects in the Phase 2/3 trial for ages 16+ had been in the trial on average for 97 days. That's 3 months plus a week.
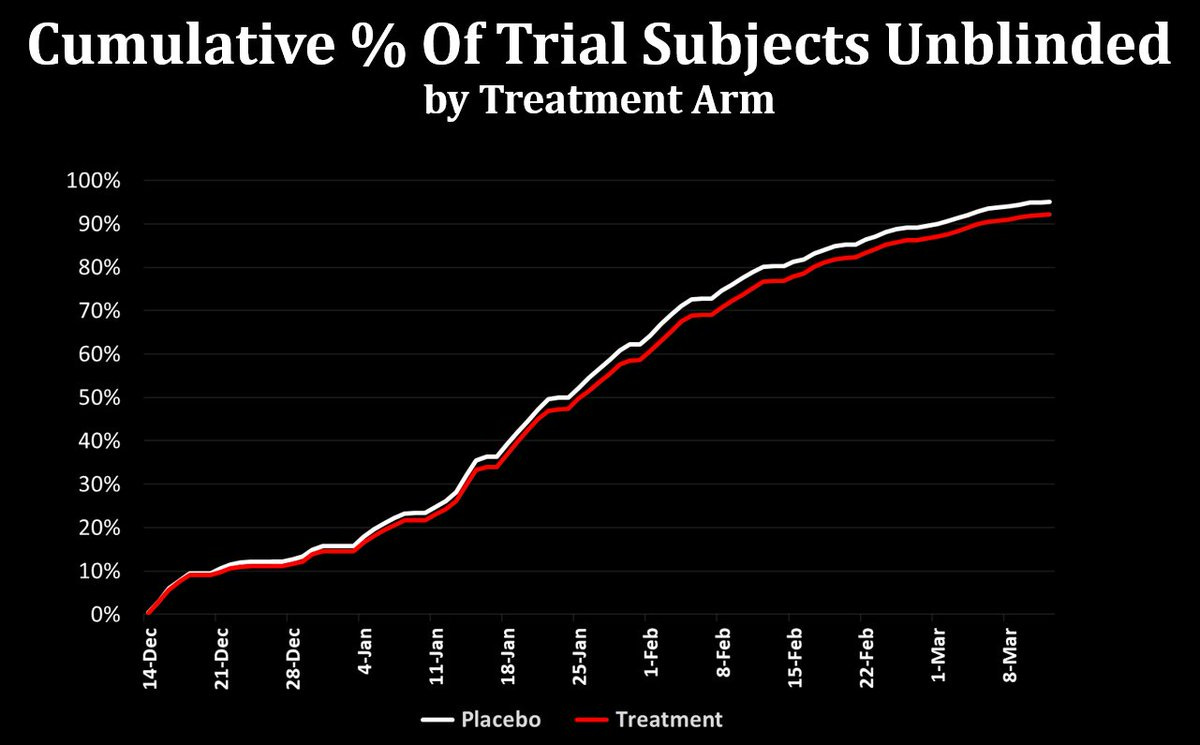
I arrived at this figure by analyzing the Pfizer/BioNTech clinical trial data released via court order, specifically using the file at this link. The figure below shows the pace of unblinding across treatment arms from Dec 14 - Mar 13. (March 13 was the data cutoff date for Pfizer’s FDA biological license application for full approval, so any trial data after that is not included in the court order.)
The first subjects recruited to phase 2/3 were given their first dose on July 27, 2020. From then to Dec. 14 is 140 days, or over 4 1/2 months. But many subjects were recruited long after July 27. In fact, 19 subjects had their first dose after Dec. 14!
But hold on a second -- they weren't all unblinded on Dec 14. Shouldn't we count how long each person was in the trial before being unblinded? If you count that way, the average subject was in the trial for 137 days before unblinding.1
Except the only way that could make sense is if the unblinding was random. If it wasn't, then the moment you start unblinding, you can't calculate an unbiased treatment effect by comparing treatment and control groups -- in other words, the conditions necessary for a Randomized Controlled Trial (RCT) no longer hold.
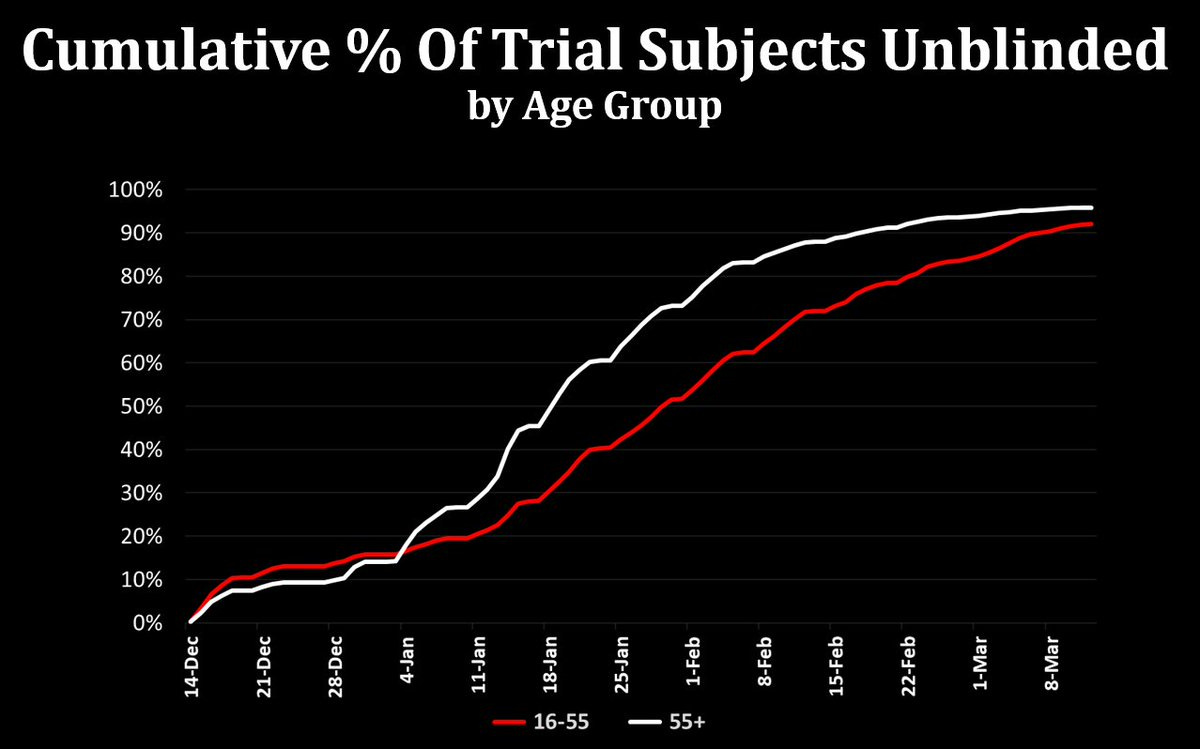
And indeed, the unblinding was not random: The Pfizer trial unblinded younger and older volunteers at different rates - not randomly!
This means the trial effectively ended on Dec 14, 2020, and we're back where we started: Subjects were in the trial an average of 97 days.
To put a little more meat on this: in the dataset linked above, there are 22,015 placebo subjects and 22,024 subjects in the treatment group. By the March 13 cutoff, 20,935 of the placebo subjects had been unblinded, with some 19,605 of them (89%) having received their first real dose of the vaccine before the data cutoff — on average 6 days after unblinding. Of those in the treatment group, 20,400 had been unblinded by the data cutoff date.
There were actually 19 subjects recruited after December 14, with the last one receiving the first dose on Jan. 8. If we set those aside, the minimum number of days in trial prior to Dec 14 was 3 days and maximum was 120, with a standard deviation of 25 days.
Once the trial effectively ended on Dec. 14, accurate assessment the safety of the vaccine across treatment and control groups was no longer possible—especially since the placebo subjects started receiving their first real dose of the vaccine very soon after unblinding. (And for more on how the trial was never double blind to begin with, see my pose here.)
[Update: I should point out that, technically, the trial is still ongoing with a completion date of May 24, 2024. Which is why I said the trial effectively ended on Dec. 14, by which I meant the randomized controlled trial. Now it’s just an open-label study with a very small and highly selective placebo group — those remaining who haven’t been vaccinated. I don’t know what kind of data they are even gathering on the trial participants, but it isn’t much.]
There were 39 placebo subjects from Phase 1 who may have been grandfathered into the 2/3 trial, this analysis excludes them. My tweet about this from a couple days ago and Jason Hart’s post about it included those subjects, which accounts for any discrepancy between this post and those. Also in my tweet, I didn’t take the space to mention the subjects who had their first dose after Dec. 14.




I don't even know if they tracked AE's and deaths that long after vaccination.... there is something about it in the trial protocol but I don't remember off the top of my head.
Right. Thank you for explaining.
You might be interested to know that the Australian Health Minister outright called the rollout “the largest global vaccination trial ever.” Sounds about right.
https://rebekahbarnett.substack.com/p/the-largest-global-vaccination-trial